Origin of Lemon Fresh Fragrance

The fragrance of this bath bomb has unique lemon scent which can be synthesized with the use of scented esters. Esters are chemical compounds that are created through a series of reactions involving alcohols and carboxylic acids through a process called esterification.
But do not worry the ester used is perfectly harmless providing a fresh lemon scent for your kids!
But do not worry the ester used is perfectly harmless providing a fresh lemon scent for your kids!
The ester used in the fragrance to give the soap bomb a lemon fresh scent is called an Ethyl butanoate.
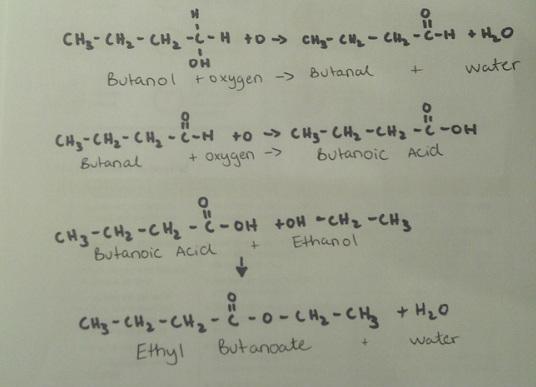
To create this ester we first start with a primary alcohol called butanol and react it with oxygen in a process called oxidation. In this reaction the oxygen binds with the two hydrogen in butanol creating water and causing a double bond between the oxygen and carbon creating a aldehyde. This aldehyde called butanal is reacted again with oxygen through oxidation adding a oxygen to the molecular compound creating a carboxyl group making butanoic acid(carboxylic acid). This carboxylic acid, butanoic acid, is then reacted with ethanol( An alcohol) through a process called esterfication to create ethyl butanoate and water as a by product.
To create this ester we first start with a primary alcohol called butanol and react it with oxygen in a process called oxidation. In this reaction the oxygen binds with the two hydrogen in butanol creating water and causing a double bond between the oxygen and carbon creating a aldehyde. This aldehyde called butanal is reacted again with oxygen through oxidation adding a oxygen to the molecular compound creating a carboxyl group making butanoic acid(carboxylic acid). This carboxylic acid, butanoic acid, is then reacted with ethanol( An alcohol) through a process called esterfication to create ethyl butanoate and water as a by product.